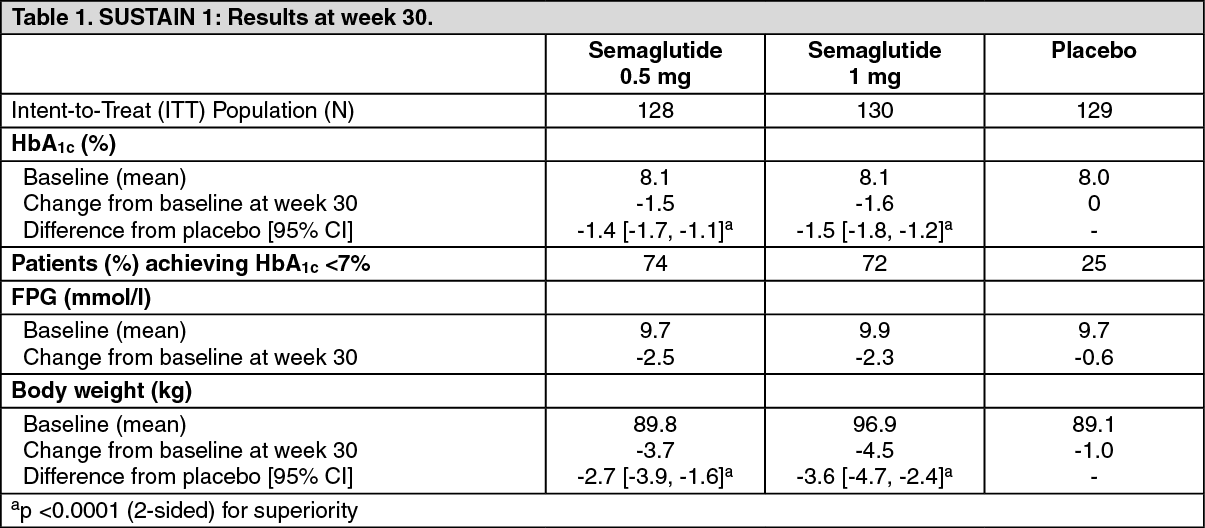
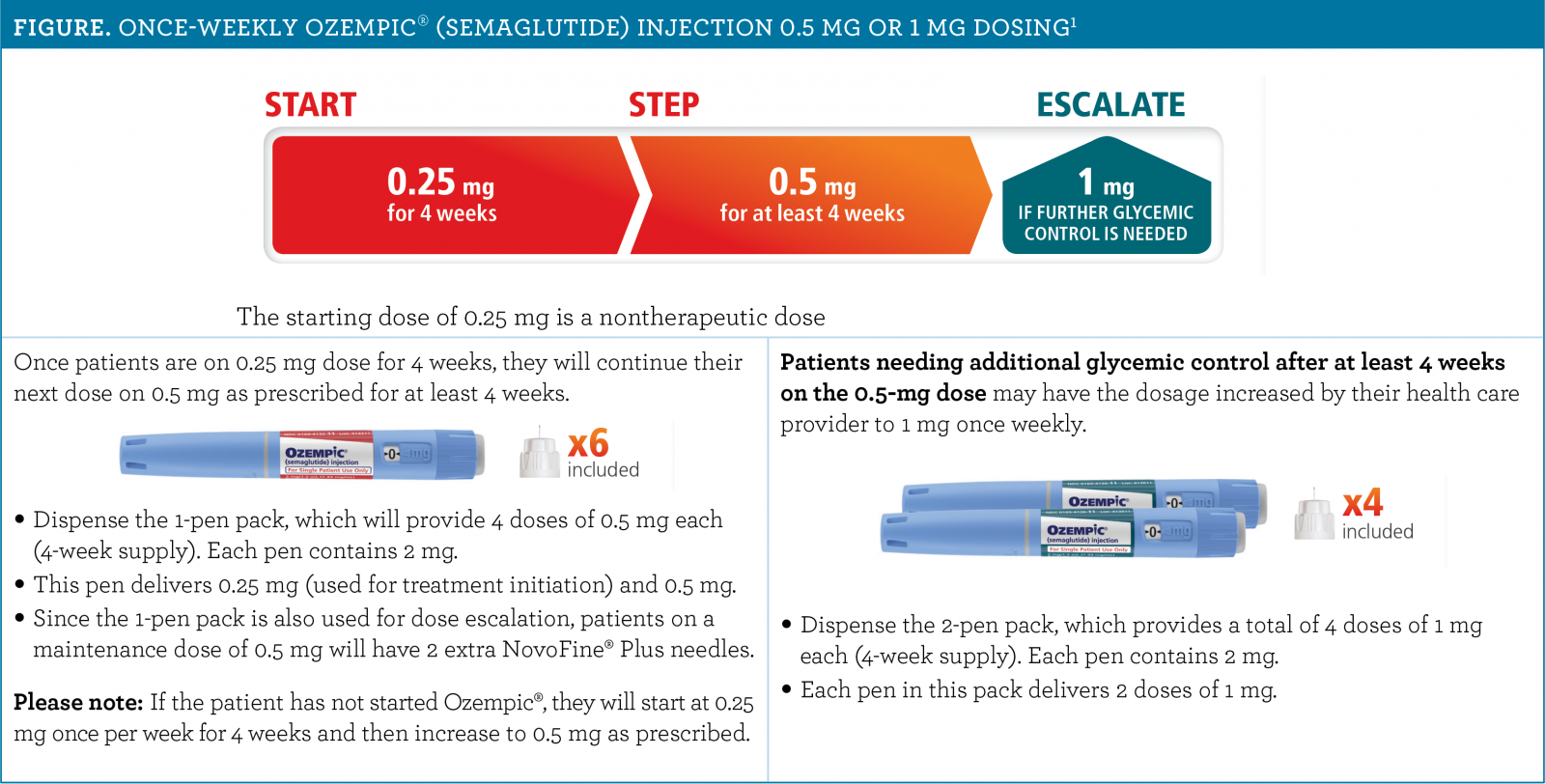
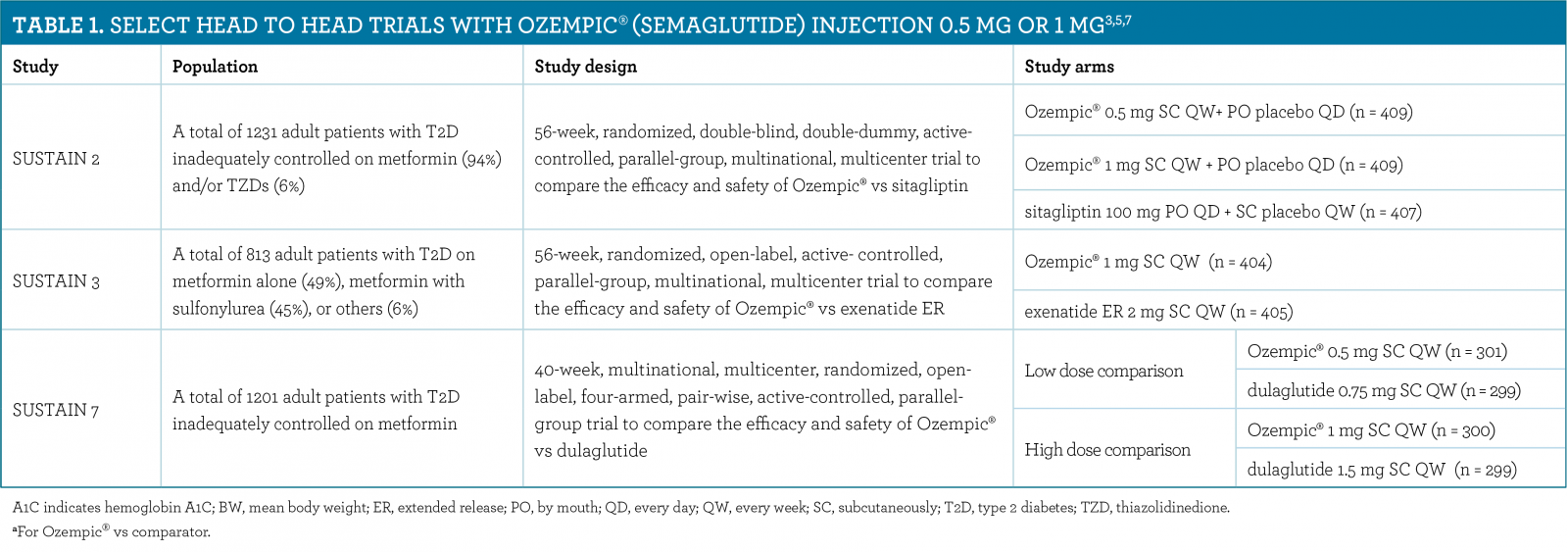
These highlights do not include all the information needed to use OZEMPIC ® safely and effectively. See full prescribing information for OZEMPIC. OZEMPIC (semaglutide) injection, for subcutaneous use Initial U.S. Approval: 2017

Ozemipic's new indication, what does it mean for type 1 diabetes management? | Integrated Diabetes Services

2nd time's the charm? Novo Nordisk reups bid for high-dose Ozempic after March FDA snub | FiercePharma

Ozempic 0.5 mg solution for injection in pre-filled pen - Summary of Product Characteristics (SmPC) - (emc)